Wearable Seismocardiography‐Based Assessment of Stroke Volume in Congenital Heart Disease
Executive Summary
This study explores the feasibility of using a multimodal wearable biosensor combining seismocardiography (SCG) and ECG to estimate stroke volume (SV) in patients with congenital heart disease (CHD). Using ridge regression and features derived from systolic time intervals, the wearable achieved an acceptable SV estimation error of 28% compared to the gold standard cardiac magnetic resonance imaging (CMR), with an R2 of 0.76. The findings suggest potential for continuous, noninvasive monitoring of cardiac function in CHD patients, though further validation in larger populations is needed.
Answer Machine Insights
Q: How accurate is the wearable biosensor in estimating stroke volume?
The wearable biosensor achieved a 28% error compared to CMR, meeting cardiac output measurement guidelines.
The percent error, the metric that is used by cardiac output measurement guidelines, for this highest performing model was 28%, within the acceptable criteria of 30%.
Q: What features were most important for stroke volume estimation?
Heart rate (HR), pre-ejection period (PEP), and ventricular ejection time (VET) were the most critical features.
Other than HR, the most important features are either the PEP, VET, or a ratio derived from the combination of them.
Key Results
Wearable biosensor achieved 28% error in SV estimation compared to CMR.
R2 correlation of 0.76 and RMSE of 11.48 mL in held-out test set.
Visual Evidence
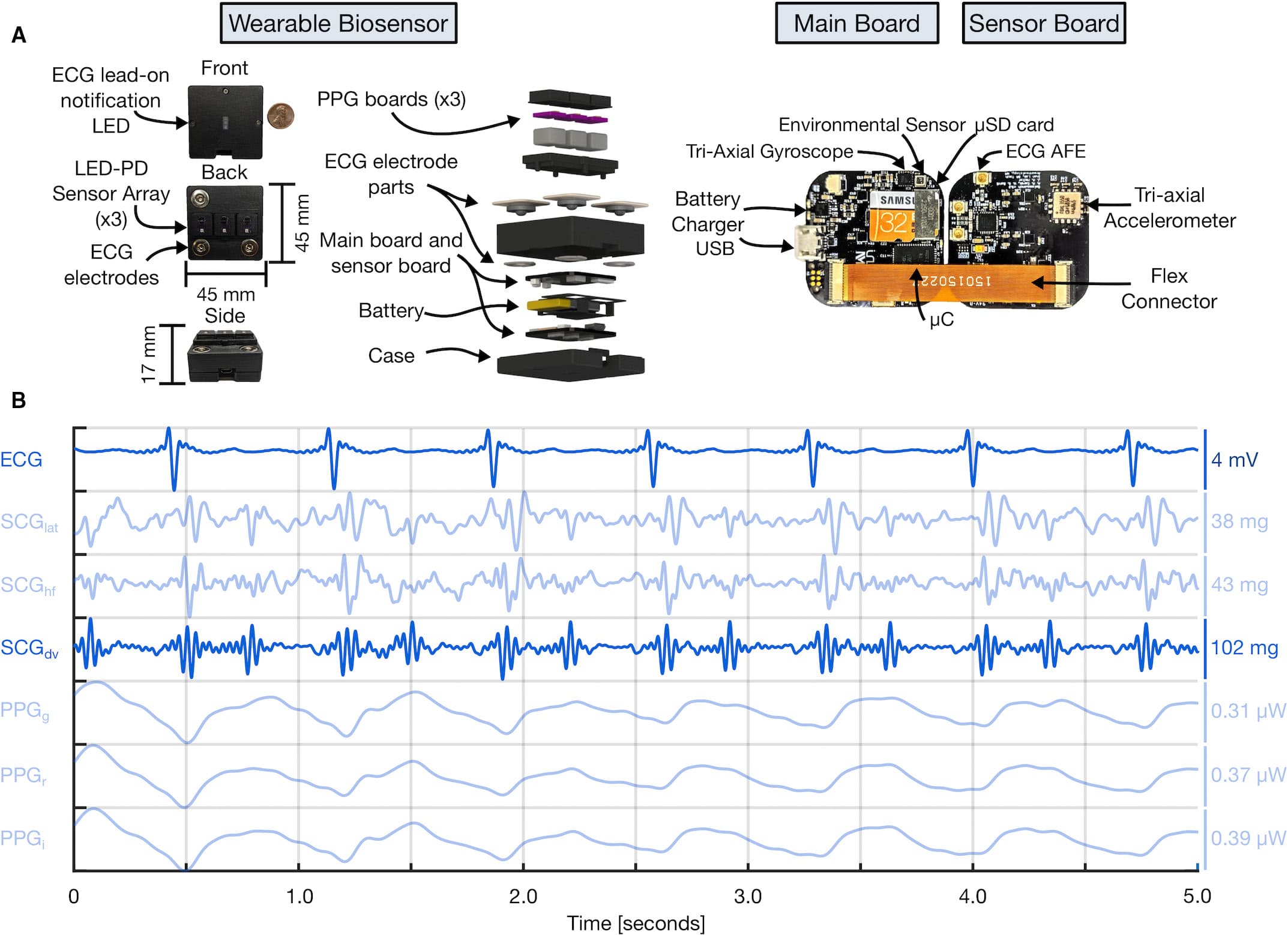
Figure 2. Wearable multimodal hardware engineering mechanics. A, Pertinent multimodal hardware diagram. Final wearable biosensor iteration with exploded view detailing photoplethysmogram (PPG) components, gel-electrode ECG connectors, lithium-polymer battery, and printed circuit boards (PCBs). Main PCB with ATSAM4LS8 microcontroller (μC), BMG250 triaxial gyroscope and BME280 environmental sensor, micro secure digital card (μSD), and BQ24232 battery charger. Sensor PCB, connected to main PCB via flexible connector, with ADXL355 accelerometer, ADS1291 analog front end, and magnetic wire connections to separate PCB containing SFH7016 multichip light-emitting diode (LED) and SFH 2703 photodiode (PD) used to acquire triaxial seismocardiogram (SCG), single-lead ECG, and multiwavelength sternum PPG signals, respectively. B, Sample 5 seconds of filtered wearable signal data from a single-ventricle patient with corresponding amplitudes are shown. In order from top to bottom: ECG, lateral SCG (SCGlat), head-to-foot SCG (SCGhf), dorso-ventral SCG (SCGdv), green PPG (PPGg), red PPG (PPGr), and infrared PPG (PPGi) signals. The darker blue ECG and SCGdv signals are those used in this work. USB indicates universal serial bus.
Clinical Snapshot
Evidence Rating
Relevance
high Priority